 Oxygen and sulfur are non-metals their elemental form is molecular, and they can gain two electrons to form ions with a two minus charge. Properties for Group 16 of Periodic Table elements They are oxygen (O), sulfur (S), selenium (Se), tellurium (Te), radioactive polonium (Po), and synthetic ununhexium (Uuh). Bismuth is also the heaviest completely stable element that does not decay radioactively to other simpler elements.Bismuth can lose three electrons to form an ion with a three-plus charge. Arsenic and Antimony are metalloids, and bismuth is the only metal in the group. Commercially, these elements are important for fertilizers. Nitrogen comprises about 3 percent of the weight of the human body and phosphorous about 1.2 percent. Nitrogen, as a diatomic molecule is the major constituent of air and both elements are essential for life.They can gain three electrons to form fairly unstable ions with a three minus charge, the nitride and phosphide ions. These elements all have five valence electrons.Properties for Group 15 of Periodic Table elements It consists of the elements Nitrogen (N), Phosphorus (P), Arsenic (As), Antimony (Sb), and Bismuth (Bi) The collective name pnictogens is also sometimes used for elements of this group. The Nitrogen group is the series of elements in Group 15 (formerly Group V) of the periodic table. The simplest oxide, silicon dioxide (SiO2) or silica, is a component of many rocks and minerals.Silicon is the second most abundant element in the earth’s crust (25.7 percent) and we are surrounded by silicon-containing materials: bricks, pottery, porcelain, lubricants, sealants, computer chips, and solar cells. Silicon in some respects is similar to carbon in that it forms four covalent bonds, but it does not form a wide range of compounds.In the elemental state, it has several forms, the most known of which are graphite and diamond. Carbon forms four covalent bonds in compounds rather than forming monatomic ions.The two metals, tin, and lead, are unreactive metals and both can form ions with a two-plus or a four-plus charge in ionic compounds.The common characteristic is four valence electrons. This group has a mixed type of element with non-metal carbon, two metalloids, and two metals.Properties of Group 14 of Periodic Table Elements Group 14 of the Periodic Table consists of the elements carbon (C), silicon (Si), germanium (Ge), tin (Sn), & lead (Pb) Aluminum is an active metal, but the stable oxide forms a protective coating over the metal making it resistant to corrosion. Aluminium is the third most abundant element in the earth’s crust (7.4 percent), and is widely used in packaging materials.The metals can lose all three electrons to form ions with a three-plus charge in ionic compounds. These elements are characterized by having three valence electrons.Boron is a metalloid, it has characteristics intermediate between metals and non-metals, and the rest of the group are metals. 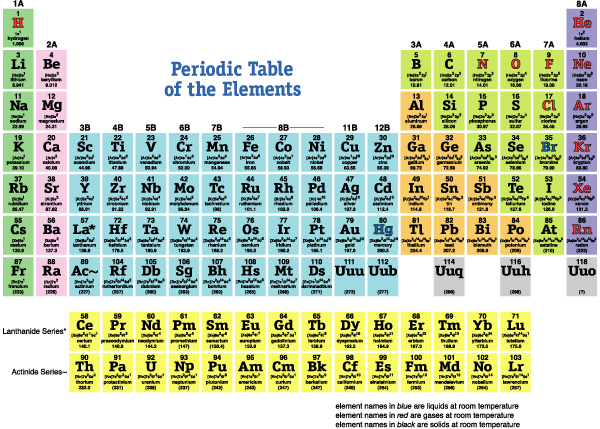
In this group, we begin to see the changeover toward the non-metallic character.Properties for Group 13 of Periodic Table elements Group 13 of the Periodic Table consists of the elements boron (B), aluminum (Al), gallium (Ga), indium (In), and thallium (Tl). They react readily with halogens to form ionic salts and can react slowly with water.Beryllium is the least metallic element in the group and tends to form covalent bonds in its compounds.These elements all have two valence electrons and tend to lose both to form ions with a two-plus charge.The alkaline earth metals are silvery-coloured, soft, low-density metals, though are a bit harder than the alkali metals.Properties for Group 2 of Periodic Table Elements Group 2 of Periodic Table The series consists of the elements beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra). They react with water to liberate hydrogen gas.Īlkali metal + water → Alkali metal hydroxide + hydrogen.The alkali metals react readily with halogens to form ionic salts, such as table salt, and sodium chloride (NaCl).Due to their activity, they occur naturally in ionic compounds, not in their elemental state.This makes them very reactive and they are the most active metals. They have the lowest ionization energies in their respective periods.These elements all have one valence electron which is easily lost to form an ion with a single positive charge.The alkali metals are silver-colored (cesium has a golden tinge), soft, low-density metals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
